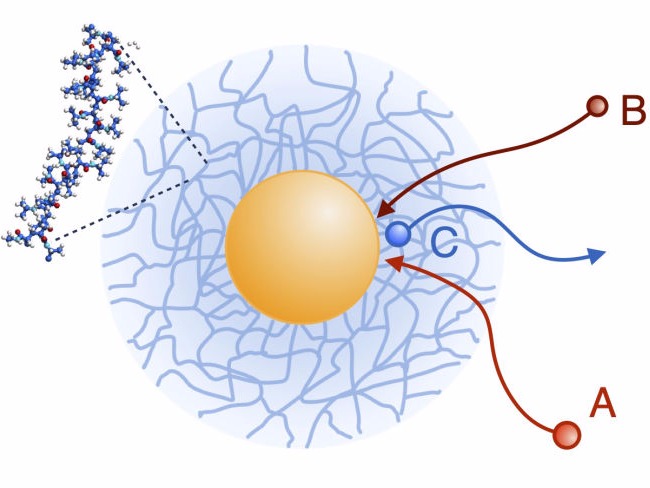
Nanoparticle catalysis: reaction-diffusion modeling
Catalysis with metallic nanoparticles is one of the most active fields in modern nanoscience. These nanoparticles have problems of aggreation, oxidation, or removal from the liquid phase after use. To solve this problem, colloidal carrier systems have been designed that encapsulate and stabilize nanoparticles by keeping them dispersed in solution. In these systems, the permeability of reactants and products in the carrier system can significantly alter the reaction rate. Using multiscale modeling, we include the molecular and polymeric structural effects of the carrier systems in advanced reaction-diffusion equations to predict reactions rates.
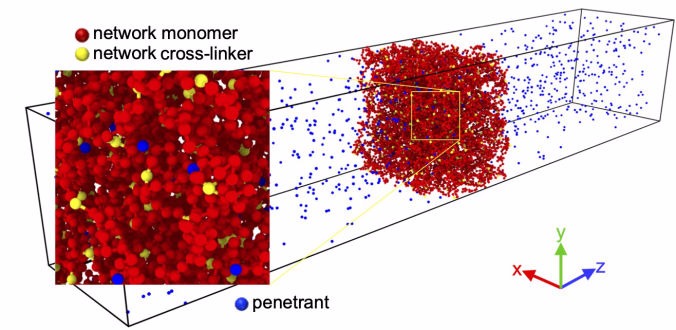
Transport phenomena in hydrogels
Hydrogels are made up of cross-linked hydrophilic polymeric chains that do not dissolve in water. Their properties can be easily adjusted by external stimuli such as pH or temperature, making them useful systems for controlled drug release, tissue engineering, biosensors, or artificial enzymes. Drawing on methods such as density functional theory, statistical mechanics, and liquid state theory, as well as coarse-grained and atomistic simulations, we analyze the diffusion, partitioning, and permeability of ions and complex molecules in hydrogels.
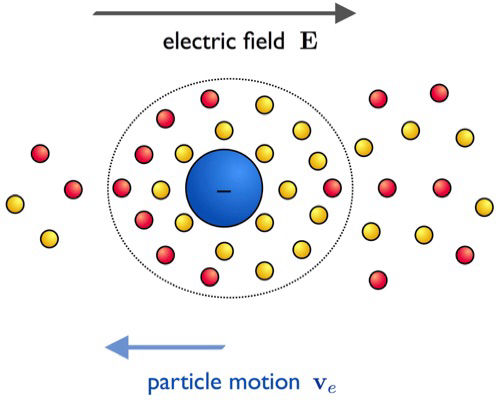
Electrostatic interactions in soft matter
Electrostatic interactions often play a crucial role when dealing with charged objects in biological and soft matter. In aqueous media, charges tend to dissociate and affect most of the functional, structural and dynamical properties of these systems. We analyze the effect of electrostatic interactions in polymer chains, synthetic polyelectrolytes, and charged colloidal particles. We are particularly interested in the theoretical study of the electric double layer and electrokinetic phenomena.
